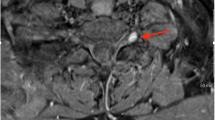
Abstract
Rasmussen’s syndrome (RS) is a rare acquired progressive inflammatory encephalopathy characterized by drug-resistant partial seizures and cognitive deterioration resulting from a gradual impairment and a subsequent atrophy of a single brain hemisphere. It was firstly described by Theodore Rasmussen in 1958. The original etiopathogenic hypothesis of a chronic viral infection no longer holds. Today, the presumed mechanism is a complex autoimmune process comprising an active role of both cytotoxic T lymphocytes and circulating autoantibodies, activating the subunit 3 of the ionotropic glutamate receptor (GluR3Ab). Several medical options, such as antiepileptic drugs and immunomodulatory therapies, deserve consideration to face epileptic manifestations and to slow neurologic regression. Yet, all these therapies turn out to be almost always just temporizing measures. Surgical disconnection of the affected hemisphere (“functional hemispherectomy”) is the best therapeutic choice to achieve the arrest of the disease, a good seizure control, the block of neurologic decline, and the improvement of patient’s quality of life.



Similar content being viewed by others
References
Alvarez-Barón E, Bien CG, Schramm J et al (2008) Autoantibodies to Munc18, cerebral plasma cells and B-lymphocytes in Rasmussen encephalitis. Epilepsy Res 80:93–97
Antel JP, Rasmussen T (1996) Rasmussen’s encephalitis and the new hat. Neurology 46:9–11
Bahi-Buisson N, Villanueva V, Bulteau C et al (2007) Long term response to steroid therapy in Rasmussen encephalitis. Seizure 16:485–492
Bauer J, Elger CE, Hans VH et al (2007) Astrocytes are a specific immunological target in Rasmussen’s encephalitis. Ann Neurol 62:67–80
Bien CG, Bauer J, Deckwert TL et al (2002) Destruction of neurons by cytotoxic T cells: a new pathogenic mechanism in Rasmussen’s encephalitis. Ann Neurol 51:311–318
Bien CG, Elger CE, Wiendl H (2002) Advances in pathogenic concepts and therapheutic agents in Rasmussen encephalitis. Expert Opin Investig Drugs 11:981–989
Bien CG, Gleissner U, Sassen R et al (2004) An open study of tacrolimus therapy in Rasmussen encephalitis. Neurology 62:2106–2109
Bien CG, Granata T, Antozzi C et al (2005) Pathogenesis, diagnosis and treatment of Rasmussen encephalitis: a European consensus statement. Brain 128:454–471
Bien CG, Widman G, Urbach H et al (2002) The natural history of Rasmussen’s encephalitis. Brain 125:1751–1759
Browner N, Azher SN, Jankovic J (2006) Botulinum toxin treatment of facial myoclonus in suspected Rasmussen encephalitis. Mov Disord 21:1500–1502
Carreño M, Donaire A, Barceló MI et al (2007) Parry Romberg syndrome and linear scleroderma in coup de sabre mimicking Rasmussen encephalitis. Neurology 68:1308–1310
Dabbagh O, Gascon G, Crowell J, Bamoggadam F (1997) Intraventricular interferon-alpha stops seizures in Rasmussen’s encephalitis: a case report. Epilepsia 38:1045–1049
Dubeau F, Andermann F, Wiendl H, Bar Or A (2008) Rasmussen’s Encephalitis (chronic focal encephalitis). In: Engel J Jr, Pedley TA (eds) Epilepsy: a comprehensive textbook. Lippincott-Williams and Wilkins, Philadelphia, pp 2439–2453
Freeman JM (2005) Rasmussen’s sindrome: progressive autoimmune multi-focal encephalopathy. Pediatr Neurol 32:295–299
Ganor Y, Goldberg-Stern H, Amrom D et al (2004) Autoimmune epilepsy: some epilepsy patients harbor autoantibodies to glutamate receptors and dsDNA on both sides of blood-brain barrier, which may kill neurons and decrease in brain fluids after hemispherotomy. Clin Develop Immunol 11:241–252
Gahring L, Carlson NG, Meyer EL, Rogers SW (2001) Granzyme B proteolysis of a neuronal glutamate receptor generates an autoantigen and is modulated by glycosylation. J Immunol 166:1433–1438
Geller E, Faerber EN, Legido A (1998) Rasmussen encephalitis: complementary role of multitechnique neuroimaging. Am J Neuroradiol 19:445–449
Granata T, Fusco L, Gobbi G et al (2003) Experience with immunomodulatory treatments in Rasmussen’s encephalitis. Neurology 61:1807–1810
Granata T, Gobbi G, Spreafico R et al (2002) Rasmussen’s encephalitis: early characteristics allow diagnosis. Neurology 60:422–425
Hart Y (2004) Rasmussen’s encephalitis. Epileptic Disord 6:133–144
Hart YM, Anderman F, Fish DR et al (1997) Chronic encephalitis and epilepsy in adults and adolescents. Neurology 48:418–424
Jonas R, Nguyen S, Hu B et al (2004) Cerebral hemispherectomy: hospital corse, seizure, developmental, language, and motor outcomes. Neurology 62:1712–1721
Leach JP, Chadwich DW, Miles JB, Hart IK (1999) Improvement in adult-onset Rasmussen’s encephalitis with long-term immunomodulatory therapy. Neurology 52:738–742
Lousa M, Sanchez-Alonso S, Rodriguez-Diaz R, Dalmau J (2000) Status epilepticus with neuron-reactive serum antibodies: response to plasma exchange. Neurology 54:2163–2165
Mantegazza R, Bernasconi P, Baggi F et al (2002) Antibodies against GluR3 peptides are not specific for Rasmussen’s encephalitis but are also present in epilepsy patients with severe, early-onset disease and intractable seizures. J Neuroimmunol 131:179–185
Mc Knight K, Jiang Y, Hart Y et al (2005) Serum antibodies in epilepsy and seizure-associated disorders. Neurology 65:1730–1736
Power C, Poland SD, Blume WT, Girvin JP, Rice GP (1990) Cytomegalovirus and Rasmussen encephalitis. Lancet 336:1282–1284
Rasmussen T (1978) Further observations on the syndrome of chronic encephalitis and epilepsy. Appl Neurophysiol 41:1–12
Rasmussen T, McCann W (1968) Clinical studies of patients with focal epilepsy due to “chronic encephalitis”. Trans Am Neurol Assoc 93:89–94
Rasmussen T, Olszweski J, Lloyd-Smith DL (1958) Focal seizures due to chronic localized encephalitis. Neurology 8:435–455
Rogers SW, Andrews PI, Gahring LC et al (1994) Autoantibodies to glutamate receptor GluR3 in Rasmussen’s encephalitis. Science 265:648–651
Root-Bernstein RS (1991) Cytomegalovirus and Rasmussen encephalitis. Lancet 337(8735):239–240
Schwab N, Bien CG, Waschbisch A et al (2009) CD8+ T-cell clones dominate brain infiltrates in Rasmussen encephalitis and persist in the periphery. Brain 132:1236–46
So NK, Gloor P (1991) Electroencephalographic and electrocorticographic findings in chronic encephalitis of the Rasmussen type. In: Anderman F (ed) Chronic encephalitis and epilepsy: Rasmussen’s syndrome. Butterworth-Heinemann, Boston, pp 37–45
Takahashi Y, Mine J, Kubota Y et al (2009) A substantial number of Rasmussen syndrome patients have increased IgG, CD4+ T cells, TNFalpha, and Granzyme B in CSF. Epilepsia 50:1419–1431
Thilo B, Stingele R, Knudsen K et al (2009) A case of Rasmussen encephalitis treated with rituximab. Nat Rev Neurol 5:458–462
Villani F, Pincherle A, Antozzi C et al (2006) Adult-onset Rasmussen’s encephalitis: anatomical-electrographic-clinical features of 7 Italian cases. Epilepsia 47(Suppl 5):41–46
Vinters HV, Wang R, Wiley CA (1993) Herpes viruses in chronic encephalitis associated with intractable childhood epilepsy. Hum Pathol 24:871–879
Walter GF, Renella RR (1989) Ebstein-Barr Virus in brain and Rasmussen encephalitis. Lancet 1:279–280
Watson R, Jiang Y, Bermudez I et al (2004) Absence of antibodies to glutamate receptor type 3 (GluR3) in Rasmussen encephalitis. Neurology 63:43–50
Wellard RM, Briellmann RS, Wilson JC et al (2004) Longitudinal study of MRS metabolites in Rasmussen encephalitis. Brain 127:1302–1312
Wiendl H, Bien CG, Bernasconi P et al (2001) GLUR3 antibodies: prevalence in focal epilepsy but no specificity for Rasmussen’ s encephalitis. Neurology 57:1511–1514
Wiles CM, Brown P, Chapel H et al (2002) Intravenous immunoglobulin in neurologic disease: a specialist review. J Neurol Neurosurg Psychiatry 72:440–448
Acknowledgments
We would acknowledge Professor Paola Iannetti, who has built and directed Child Neurology Division of our institution until 30/06/2009, for having inspired the realization of this manuscript.
Conflict of interest
None of the authors have any conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mastrangelo, M., Mariani, R. & Menichella, A. Eponym. Eur J Pediatr 169, 919–924 (2010). https://doi.org/10.1007/s00431-010-1148-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-010-1148-0